|
9/25/2023 0 Comments Rheed vs xray diffraction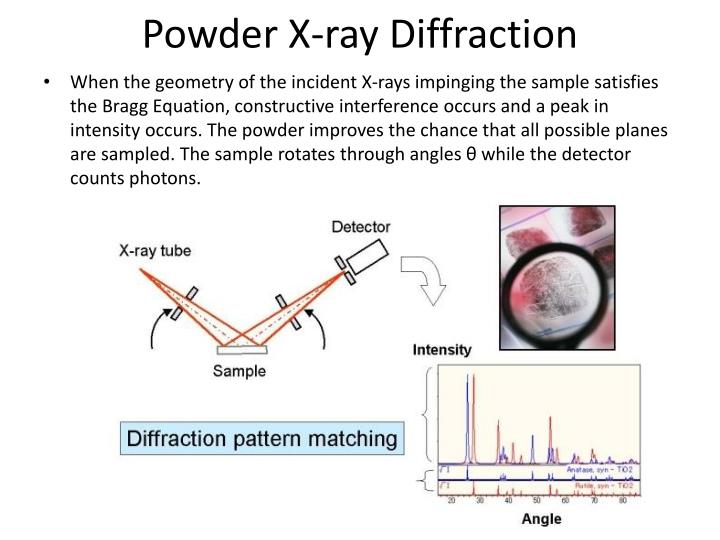
You can change three variables (d, λ, and θ) to see how they effect the diffraction. When the meter is green it indicates that Bragg’s law is satisfied. If you click on the details button you can see the detector, which measures how well the phases of the two rays match. Bragg’s Law is satisfied and diffraction is occurring. At the beginning the scattered rays are in phase and interfering constructively. Guide to how to use Applet: There are 2 rays incident on two atomic layers of a crystal (d). N = integer representing the order of the diffraction peak.ĭ = inter-plane distance of (i.e atoms, ions, molecules)Ĭlick on the following image below to get to an Applet where you can explore this relationship of Bragg’s Law Lawrence Bragg and is known as Bragg’s Law The relationship describing the angle at which a beam of X-rays of a particular wavelength diffracts from a crystalline surface was discovered by Sir William H. Available here Image Courtesy:ġ.”Freezed XRD”By Kaspar Kallip – Own work, (CC BY-SA 4.0) via Commons WikimediaĢ.”DifraccionElectronesMET”By Oysteinp at the English language Wikipedia, (CC BY-SA 3.\( \newcommand\) “Electron Diffraction.” Wikipedia, Wikimedia Foundation, 12 July 2018. (a) X-ray diffraction L scan about the STO 002 peak (b) H scan about the STO 200. “X-Ray Crystallography.” Wikipedia, Wikimedia Foundation, 7 Nov. Download scientific diagram Typical x-ray diffraction and RHEED data used to determine STO lattice constants. The key difference between X ray diffraction and electron diffraction is that X ray diffraction involves the diffraction of an incident beam of X rays into different directions whereas electron diffraction involves the interference of an electron beam. 
Summary – X Ray Diffraction vs Electron Diffractionīoth X ray diffraction and electron diffraction are techniques that we can use to determine the structure of crystals. More details are shown in the infographic on difference between X ray diffraction and electron diffraction. As another important difference between X ray and electron diffractions, electron diffraction is limited by phase problem while it has no considerable effect on X ray diffraction. Moreover, X ray diffraction uses a beam of X rays while electron diffraction uses a beam of electrons. X ray diffraction and electron diffraction are important analytical techniques that we can use to determine the crystal structure of crystalline solids. What is the Difference between X Ray Diffraction and Electron Diffraction? These instruments use an accelerated electron beam (accelerated by an electrostatic potential). Usually, we perform this experiment in a transmission electron microscope (TEM) or in a scanning electron microscope (SEM). The term interference refers to the formation of a resultant wave from two waves that have greater, lower or equal amplitude. Hence, the theory behind this technique involves the firing of electrons at a sample to observe the interference patterns of the electron beam. What is Electron Diffraction?Įlectron diffraction is an analytical technique that we use to study the matter. However, these waves cancel one another via destructive interference. We call this process as “elastic scattering” and electron acts as the scatterer. As a result, X rays striking the electrons produce secondary waves (spherical waves) emerge from the electron. 
Therefore, the atoms in the crystal can scatter the X-ray beams through the electrons of the atoms. X rays are waves of electromagnetic radiation. Furthermore, we can determine the chemical bonds and various other information as well.Ĭrystals have regularly arranged atoms. Consequently, the electron densities give the positions of atoms in the crystal structure. In brief, by measuring the angles and intensities of the diffracted beams, we can determine the 3D picture of the electron density within that crystal. Hence, the theory behind the technique involves the diffraction of an incident X ray beam into different directions. X ray diffraction or X ray crystallography is an analytical technique that we use to determine the structure of crystals. Side by Side Comparison – X Ray Diffraction vs Electron Diffraction in Tabular Form Therefore the applications of these techniques are in solid-state physics and chemistry. These techniques reveal the crystal structures of matter. Another such technique is neutron diffraction. The key difference between X ray diffraction and electron diffraction is that X ray diffraction involves the diffraction of an incident beam of X rays into different directions whereas electron diffraction involves the interference of an electron beam.īoth X ray diffraction and electron diffraction are analytical techniques that we can use to study matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
